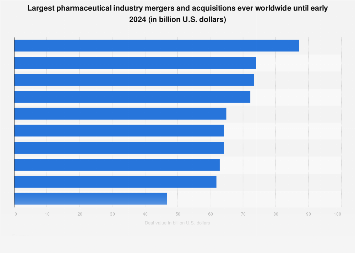
The pharmaceutical industry, a sector perpetually driven by innovation and scale, has a history punctuated by colossal mergers and acquisitions. These seismic shifts in corporate structure are not merely financial transactions; they represent strategic realignments, intensified competition, and a relentless pursuit of market dominance and scientific advancement. As of early 2024, a select group of these mega-deals stands as a testament to the industry’s immense capital flows and its ambition to shape the future of medicine. These transactions, often valued in the tens or even hundreds of billions of dollars, have fundamentally altered the competitive terrain, reshaped research and development pipelines, and influenced healthcare access and affordability on a global scale.
The sheer magnitude of these pharmaceutical mergers is a defining characteristic. When companies of such size and scope combine, the resulting entity often becomes a formidable force, capable of leveraging economies of scale, expanding its geographic reach, and integrating diverse therapeutic portfolios. These deals are typically fueled by a confluence of factors: the desire to acquire promising drug candidates in burgeoning therapeutic areas, the need to achieve cost efficiencies through consolidation, the strategic imperative to bolster market share against rivals, and the ambition to build a more diversified revenue stream capable of weathering patent cliffs and the inherent risks of drug development. The process leading up to such a monumental agreement is invariably complex, involving extensive due diligence, intricate negotiations, regulatory scrutiny from multiple jurisdictions, and significant shareholder approvals.
One of the most striking examples in recent memory, and indeed one of the largest in the industry’s annals, was the acquisition of Allergan by AbbVie. This transformative deal, valued at approximately $63 billion, was finalized in May 2020. The merger brought together AbbVie’s expertise in immunology and oncology with Allergan’s robust portfolio in eye care, neuroscience, and medical aesthetics. The strategic rationale was clear: to create a more diversified biopharmaceutical powerhouse with a broader range of revenue streams and a more resilient business model. AbbVie, already a major player with its blockbuster drug Humira, sought to reduce its reliance on a single product and expand into high-growth areas. Allergan, with its innovative pipeline and established brands, found a powerful partner to accelerate its growth and global presence.
The impact of such large-scale integrations extends far beyond the balance sheets of the involved companies. For shareholders, these mergers often promise enhanced value through synergistic cost savings and accelerated growth prospects. For employees, the integration can bring about both opportunities and challenges, with potential redundancies in overlapping functions alongside new roles in expanded divisions. Crucially, for patients and healthcare systems, the consequences are multifaceted. On the one hand, the combined entity’s increased scale and R&D capabilities could lead to the faster development of new and improved treatments. On the other hand, concerns about market concentration and potential price increases for essential medicines are frequently raised by regulators and patient advocacy groups. The regulatory review process for deals of this magnitude is typically rigorous, with antitrust authorities scrutinizing potential impacts on competition and consumer welfare.
Beyond the AbbVie-Allergan transaction, the pharmaceutical M&A landscape has witnessed other epoch-making events. The Pfizer-Wyeth merger in 2009, valued at around $68 billion, was another transformative event that significantly reshaped Pfizer’s global footprint and therapeutic offerings, bolstering its position in areas like vaccines and biosimilars. Similarly, the acquisition of Shire by Takeda Pharmaceutical in 2019, a staggering $59 billion deal, created a leading global specialty and R&D-driven biopharmaceutical company, particularly strong in rare diseases and neuroscience. These deals underscore a recurring theme: the drive to achieve critical mass, diversify risk, and gain access to cutting-edge science and profitable markets.
The market data surrounding these mega-mergers often reveals a consistent pattern. Companies typically pursue acquisitions when their internal R&D pipelines face challenges, when key drugs are approaching patent expiration, or when seeking to enter rapidly expanding therapeutic niches. For instance, the rise of biologics and gene therapies has spurred a wave of M&A activity, as established pharmaceutical giants look to acquire smaller, agile biotech firms possessing novel technologies and promising early-stage assets. The financial valuations are often driven by the projected future revenues of the acquired company’s pipeline, discounted back to present value, and augmented by potential cost synergies.
Global comparisons highlight the distinct strategies employed by pharmaceutical giants across different regions. While US and European companies have historically been at the forefront of large-scale M&A, Asian pharmaceutical firms, particularly those in China and Japan, have increasingly become active players, both as acquirers and targets, reflecting their growing R&D capabilities and ambition for global reach. The increasing complexity of drug development, coupled with the rising cost of bringing new medicines to market, incentivizes consolidation as a means to share risk and pool resources for ambitious research programs.
The economic impact of these mergers is profound. They contribute significantly to global economic activity through investment, job creation (though sometimes with associated job losses), and the stimulation of related industries such as consulting, legal services, and investment banking. Furthermore, the enhanced R&D capabilities of consolidated entities can, in the long run, lead to the development of life-saving therapies, improving public health outcomes and reducing the overall burden of disease. However, the economic benefits must be weighed against potential downsides, such as increased market power that could lead to higher drug prices, impacting affordability for patients and healthcare systems worldwide. Regulatory bodies often play a crucial role in balancing these competing interests, ensuring that mergers serve the broader public good by fostering innovation and maintaining access to medicines.
Looking ahead, the trends suggest that pharmaceutical M&A will continue to be a defining feature of the industry. The ongoing advancements in areas like artificial intelligence for drug discovery, personalized medicine, and gene editing are creating new avenues for therapeutic breakthroughs, which will inevitably lead to further strategic consolidations. Companies will continue to seek out innovative technologies and promising pipelines to maintain their competitive edge in an ever-evolving healthcare landscape. The pursuit of scale, diversification, and scientific leadership will ensure that the era of the mega-merger in pharmaceuticals is far from over, promising to continue reshaping the global health sector for years to come.