The Delhi High Court recently navigated a complex intersection of intellectual property rights, public health imperatives, and commercial viability, ultimately declining to mandate the destruction of Dr. Reddy’s Laboratories Ltd.’s existing inventory of ‘Olymviq,’ a generic version of the blockbuster diabetes drug semaglutide. This pivotal decision emerged from a trademark infringement suit initiated by Danish pharmaceutical giant Novo Nordisk A/S, the innovator behind Ozempic, a globally renowned brand of semaglutide. The court’s oral observations, particularly Justice Jyoti Singh’s emphasis on the potential harm to diabetic patients from destroying readily available medication, underscore a broader societal debate on balancing corporate brand protection with ensuring access to essential medicines in a high-demand market.
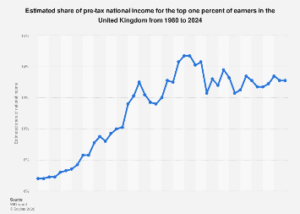
At the heart of this dispute lies semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist that has revolutionized the treatment landscape for type 2 diabetes and, more recently, weight management. Marketed by Novo Nordisk under brand names like Ozempic, Rybelsus, and Wegovy, semaglutide has achieved unprecedented commercial success, with global sales for Ozempic alone reportedly exceeding $63 billion over the past five years. Its efficacy in blood sugar control and significant weight loss has fueled immense demand, creating a lucrative, yet fiercely contested, global market. The patent exclusivity for semaglutide in India expired on March 20, triggering an anticipated rush of domestic generic manufacturers eager to tap into this high-growth segment, a phenomenon characteristic of India’s role as the "pharmacy of the world." Dr. Reddy’s entry with ‘Olymviq’ represents one such significant move to capitalize on this expiry.
Novo Nordisk lodged its complaint with the Delhi High Court on November 25, asserting that Dr. Reddy’s ‘Olymviq’ brand name bore a deceptive resemblance to its established ‘Ozempic’ trademark, potentially confusing both patients and healthcare prescribers. The Danish firm argued that ‘Ozempic’ is a coined, well-known brand enjoying global recognition and significant goodwill, and that allowing similar nomenclature in the same therapeutic class would dilute its brand equity and introduce an unacceptable risk of confusion in a critical healthcare context. In an earlier hearing, the court had indeed instructed Dr. Reddy’s to cease the manufacture and distribution of products under the ‘Olymviq’ trademark, pending further judicial review. However, the subsequent plea by Novo Nordisk for the destruction or repackaging of the already manufactured stock met with judicial resistance.
Justice Singh’s explicit observation that "destroying already manufactured stock of a diabetes drug would not be in the patients’ interest" resonates deeply within India’s public health context. With an estimated 77 million adults living with diabetes, India faces one of the highest burdens of the disease globally. The availability of affordable, effective treatments like generic semaglutide is crucial for managing this widespread chronic condition and preventing severe complications. The court’s reluctance to order destruction reflects a pragmatic approach, prioritizing patient access over strict adherence to corporate trademark enforcement in a scenario involving life-saving medication. This judicial stance aligns with a broader policy imperative in developing economies to ensure that intellectual property rights do not unduly impede access to essential medicines, especially post-patent expiry.

The complexities of the situation extend beyond mere destruction. Novo Nordisk, through its counsel Senior Advocate Amit Sibal, had also suggested that Dr. Reddy’s revert to an alternative brand, ‘Obeda,’ which it already possessed, or undertake relabelling of the ‘Olymviq’ stock. Dr. Reddy’s, represented by Senior Advocate Sandeep Sethi, countered these suggestions, highlighting the practical and commercial challenges. Sethi noted that over-labelling, especially for injectable products, could introduce risks, including potential contamination or issues with product integrity, thereby posing unforeseen patient safety concerns. Furthermore, the court itself expressed skepticism regarding the commercial viability of relabelling, questioning who would purchase a product with an altered or obscured label. These arguments underscore the operational complexities involved in remedying trademark infringements for pharmaceutical products that have already entered the supply chain.
The broader economic implications of this dispute are significant for both the Indian and global pharmaceutical industries. For Novo Nordisk, the aggressive protection of its intellectual property in emerging markets like India is vital to maintaining its competitive edge and recouping substantial R&D investments. The ‘Ozempic’ brand, along with ‘Wegovy,’ represents a cornerstone of the company’s revenue, and any perceived dilution or unconstrained generic entry could set an unwelcome precedent. For Indian generic manufacturers like Dr. Reddy’s, the opportunity to introduce affordable versions of blockbuster drugs immediately after patent expiry is a core business model. This enables them to serve a vast domestic market and export to other developing nations, solidifying India’s position as a global leader in generic drug manufacturing. The successful entry of generic semaglutide could significantly reduce treatment costs for millions of Indian patients, thereby easing the burden on individual households and the national healthcare system. Market analysts project the global GLP-1 agonist market to reach over $100 billion by the end of the decade, with a substantial portion of this growth expected from increased accessibility in emerging markets.
This case also sheds light on the evolving landscape of intellectual property law in India, particularly concerning the balance between protecting brand integrity and promoting public welfare. While trademark law primarily aims to prevent consumer confusion and protect a brand owner’s goodwill, judicial interpretations in the pharmaceutical sector often introduce a "public interest" lens. Legal experts suggest that courts are increasingly willing to consider the broader societal impact when ruling on disputes involving essential medicines. For instance, in similar cases globally, courts have often looked for "deceptive similarity" from the perspective of an average prudent consumer, but for drugs, the potential for patient confusion carries far graver consequences. The Delhi High Court’s current stance, allowing the existing stock to be released without further brand promotion while halting future manufacture under the disputed name, signals a nuanced approach: penalizing future infringement but mitigating immediate patient impact.
Dr. Reddy’s assurance to the court that it would not manufacture semaglutide under the ‘Olymviq’ trademark moving forward, coupled with its request to release the existing inventory manufactured in February, offers a potential pathway for resolution. The court’s indication that a "balanced solution" could be explored, possibly allowing the existing stock to be utilized subject to safeguards agreeable to Novo Nordisk, suggests a mediated outcome rather than an outright win for either party. This approach could involve stringent conditions on distribution, perhaps limited to specific channels or geographies, to minimize brand confusion while ensuring the medication reaches patients.
The ongoing legal skirmish between Dr. Reddy’s and Novo Nordisk is not isolated to this trademark dispute. Dr. Reddy’s had also filed trademark applications for ‘Olymviq’ in July 2025 and March 2026, indicating a strategic intent to secure the brand. Furthermore, the two pharmaceutical giants have been embroiled in a separate legal battle since May 2025 concerning alleged infringement of the semaglutide patent itself, highlighting the multi-faceted nature of competition in the high-stakes pharmaceutical sector. This complex interplay of patent and trademark litigation is expected to become a more frequent feature as innovative drugs with high commercial value reach their patent cliff, particularly in dynamic markets like India. The ultimate resolution of these disputes will not only define the market trajectory for semaglutide in India but also establish crucial precedents for brand protection and generic drug accessibility in the country’s rapidly expanding pharmaceutical landscape.