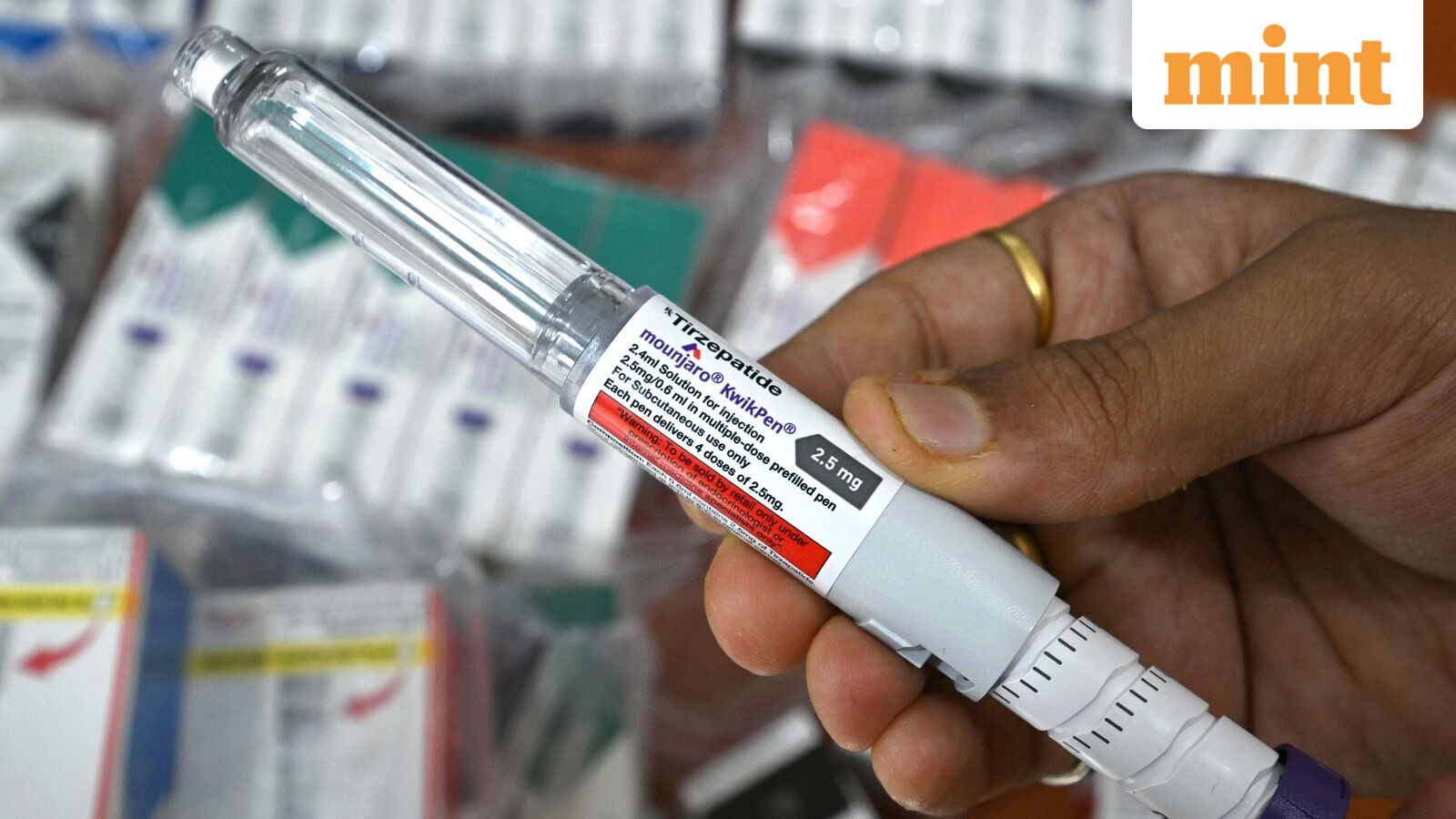
The Indian pharmaceutical landscape is undergoing a seismic shift, poised to democratize access to life-changing weight-loss and diabetes treatments following the immediate launch of generic semaglutide versions. A day after key patent protection for the blockbuster GLP-1 agonist lapsed in India, leading domestic drugmakers, including industry stalwarts Dr Reddy’s Laboratories, Sun Pharmaceutical Industries, and Zydus Lifesciences, swiftly introduced their affordable alternatives. This aggressive entry has triggered an intense price war, with generic formulations priced a staggering 50-90% below the innovator brands, fundamentally reshaping the trajectory of India’s nascent yet rapidly expanding market for these therapies.
This pivotal development marks a significant inflection point for healthcare affordability and access in a nation grappling with a burgeoning epidemic of metabolic disorders. Until now, the high cost associated with innovator GLP-1 drugs, such as Novo Nordisk’s Ozempic and Wegovy, had largely restricted their reach to an affluent segment of the population, despite overwhelming global demand for their efficacy in managing Type 2 diabetes and chronic weight. The arrival of generics promises to unlock a vast untapped market, potentially transforming public health outcomes and solidifying India’s position as a global pharmaceutical powerhouse.
Glucagon-like peptide-1 (GLP-1) receptor agonists like semaglutide operate by mimicking a natural gut hormone that plays a crucial role in glucose metabolism and appetite regulation. By signaling the brain to feel full, slowing gastric emptying, and stimulating insulin release, these drugs effectively help patients reduce food intake, achieve significant weight loss, and improve glycemic control. The global pharmaceutical industry has witnessed an unprecedented surge in demand for these "miracle drugs," with their market projected to reach hundreds of billions of dollars globally in the coming years. India, often referred to as the "pharmacy of the world," is now strategically positioned to contribute significantly to the global supply of affordable GLP-1 therapies, following the expiry of intellectual property rights that have paved the way for generic competition.
The price erosion is immediate and substantial. Novo Nordisk’s pioneering brands, Ozempic and Wegovy, which made their debut in India last year, were priced at approximately ₹8,800 and ₹10,850 per month, respectively, for starting doses delivered via convenient pen devices. In stark contrast, the newly launched generic versions offer a spectrum of pricing, ranging from as low as ₹1,300 per month for vial-based formulations to over ₹4,000 for pre-filled pen devices. This dramatic reduction is expected to dismantle a significant financial barrier for millions of Indian patients.
Leading the charge in this competitive landscape, Dr Reddy’s Laboratories launched its brand Obeda at ₹4,200 per month for 2mg and 4mg doses in a pre-filled pen device, initially indicated for Type 2 diabetes. The company has also signaled its intent to seek regulatory approval for weight management, anticipating broader utility. Sun Pharmaceutical Industries introduced two brands, Noveltreat and Sematrinity, with Noveltreat, targeting chronic weight management, priced between ₹3,600 and ₹8,000 monthly across five dose strengths. Sematrinity, for Type 2 diabetes, ranged from ₹3,000 to ₹5,200 monthly, both delivered in pre-filled pens. Zydus Lifesciences, in an aggressive move to undercut rivals, announced an average treatment cost of approximately ₹2,200 per month for its three brands – Semaglyn, Mashema, and Alterme – notably holding regulatory approval for both diabetes and weight management. Zydus has also strategically partnered with Lupin and Torrent Pharma for broader distribution. Meanwhile, Glenmark Pharmaceuticals’ Glipiq, available in vial form for Type 2 diabetes, offers even lower monthly costs of ₹1,300 to ₹1,760, with a pen-device version expected soon. Natco Pharma, arguably firing the initial salvo, debuted its brands Semanat and Semafull at an ultra-competitive ₹1,290 per month in vial form, with a pen device anticipated next month priced around ₹4,000-₹4,500. Eris Lifesciences, through its partnership with Natco, is offering its Sundae brand at identical starting prices. This intense competition underscores the massive market opportunity and the fierce battle for market share.
The impetus behind this accelerated generic rush is rooted in compelling market dynamics. In just one year since the introduction of innovator GLP-1 brands like Mounjaro and Wegovy, the Indian market has swelled to over ₹1,400 crore. CareEdge Ratings projects an astonishing fivefold expansion of this market within the next five years, signaling robust growth potential. Further analysis by Systematix Institutional Equities estimates the generic semaglutide opportunity across India, emerging markets, and regulated markets to exceed ₹5,000 crore over the next 12-15 months alone. This substantial pie is expected to be carved up among 10-15 global and Indian generic manufacturers. Looking further ahead to fiscal year 2027, the report forecasts an incremental revenue opportunity of ₹1,000-₹2,000 crore within India’s branded formulations market, a formidable ₹4,500 crore in regulated markets such as Canada and Brazil, and an additional ₹500-₹1,000 crore in other emerging economies.
India’s pharmaceutical manufacturing prowess, characterized by its cost-efficiency, advanced R&D capabilities, and stringent quality controls, positions it uniquely to capitalize on this global patent cliff. With semaglutide losing patent exclusivity in over 80 countries this year, Indian drugmakers like Dr Reddy’s, Sun Pharma, Biocon, and Ajanta Pharma are strategically targeting both domestic and international markets, reinforcing India’s role as the "pharmacy of the developing world" and an increasingly significant player in regulated markets. This global generic opportunity not only promises substantial revenue streams for Indian companies but also heralds a new era of affordability in global healthcare.
Beyond the immediate financial gains and market share battles, the generic GLP-1 revolution carries profound public health implications for India. The country faces an alarming increase in non-communicable diseases, with diabetes and obesity rates soaring across urban and even rural populations. According to the International Diabetes Federation, India is home to the second-highest number of people with diabetes globally, and obesity prevalence continues to rise steadily. The prohibitive cost of innovator drugs meant that only a fraction of those who could benefit from GLP-1 therapies had access. The availability of significantly cheaper generics is expected to dramatically widen treatment accessibility, potentially leading to better disease management, reduced complications, and improved quality of life for millions. This wider access could also alleviate the long-term burden on the public healthcare system by preventing costly complications associated with uncontrolled diabetes and obesity.
The strategic vision of Indian pharmaceutical companies extends beyond just the pill. Dr Reddy’s, for instance, is already planning consumer-focused complementary offerings through its joint venture with Nestle. The forthcoming launch of "Celevida GLP+" in April, a product specifically designed to support patients on GLP-1 therapy, alongside new diabetic biscuits with a lower glycemic index, illustrates a growing trend towards holistic patient care. This integrated approach, combining pharmacotherapy with nutritional support and lifestyle management, is critical for maximizing therapeutic outcomes and promoting sustainable health improvements. Such initiatives highlight a maturing market that understands the importance of a comprehensive ecosystem for managing chronic conditions.
However, the widespread availability of affordable GLP-1 generics also presents challenges. Ensuring appropriate prescription, preventing misuse (especially for purely cosmetic purposes), and educating both healthcare professionals and the public about the drugs’ benefits and potential side effects will be paramount. Regulatory bodies will need to remain vigilant to ensure quality and safety standards are maintained across the multitude of generic offerings. Furthermore, the intense competition might squeeze profit margins in the long run, necessitating continuous innovation and differentiation strategies from manufacturers.
In conclusion, the generic semaglutide launches in India represent more than just a fierce price war; they signify a fundamental reorientation of the global pharmaceutical landscape and a monumental stride towards healthcare equity. By leveraging its robust manufacturing capabilities and strategic market timing, India is not only addressing its own pressing public health challenges but also positioning itself as a pivotal provider of affordable, life-altering medicines to the world. This moment could well define the next chapter for India’s pharmaceutical industry, promising both significant economic gains and transformative health benefits on a global scale.