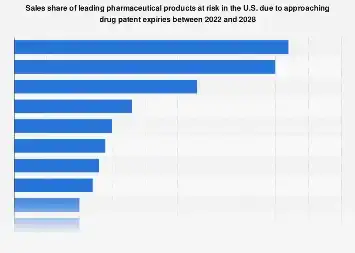
The pharmaceutical industry is bracing for a significant shift in market dynamics as a wave of blockbuster drugs approaches patent expiry, creating a substantial risk to current sales shares and signaling a fertile ground for generic competition. This impending patent cliff represents not merely a challenge but a fundamental reshaping of the competitive landscape for many leading drug manufacturers, particularly within the United States, a market that often dictates global trends. The expiration of exclusivity rights for a number of high-revenue-generating medications is expected to usher in an era of intensified price competition and a redistribution of market dominance.
For years, pharmaceutical companies have invested heavily in research and development, securing market exclusivity for their innovative therapies through patents. This period of patent protection allows them to recoup their substantial R&D costs and generate significant profits, often reaching billions of dollars annually for a single drug. However, the finite lifespan of these patents means that once they expire, the market opens up to lower-cost generic alternatives. This transition can lead to a rapid and dramatic erosion of sales for the originator drug, as healthcare providers and patients opt for more affordable versions.
The United States, with its vast healthcare market and robust regulatory framework for drug approval, is a particularly critical battleground for this patent cliff phenomenon. Data analysis indicates that a substantial portion of the market share currently held by these originator drugs is vulnerable. While specific figures can fluctuate based on market access, physician prescribing habits, and the efficacy of generic alternatives, the potential for revenue loss is universally acknowledged as a major concern for the sector. Industry analysts consistently point to the period following patent expiry as a time of acute financial pressure for companies reliant on these blockbuster products.
The economic implications of this shift are far-reaching. For pharmaceutical giants, the loss of exclusivity for even one major drug can translate into billions of dollars in lost annual revenue. This can impact stock valuations, R&D budgets for future innovations, and overall corporate strategy. Companies that have not adequately diversified their product portfolios or developed robust pipelines of new, patent-protected drugs are particularly exposed. This has spurred a strategic re-evaluation within many organizations, focusing on mergers and acquisitions, licensing agreements, and the accelerated development of next-generation therapies to mitigate the impact of expiring patents.
Conversely, the generic pharmaceutical sector stands to benefit immensely. Companies specializing in the production of generic drugs are well-positioned to capitalize on these market openings. The lower cost of development and manufacturing for generic versions, coupled with established distribution channels, allows them to offer significantly cheaper alternatives. This not only increases their market share but also contributes to broader healthcare cost containment efforts. Regulatory bodies often expedite the approval process for generic drugs once patents expire, further accelerating the market entry of these more affordable options.
The competitive dynamics at play are complex. While the price differential between branded and generic drugs is often substantial, factors such as physician preference, patient adherence, and the perceived superiority of the branded drug can influence the speed and extent of market share erosion. Some originator companies attempt to mitigate these losses through strategies like authorized generics, where they themselves launch a generic version of their own drug, or by developing "supergenerics" with minor improvements or different delivery mechanisms that may extend some form of market exclusivity. However, these strategies often provide only a temporary reprieve.
Globally, the impact of patent expiries is a recurring theme in the pharmaceutical industry. While the U.S. market is a significant driver, similar trends are observed in Europe, Japan, and other major pharmaceutical markets. The timing of patent expiries, however, can vary by region due to differences in patent laws and regulatory approval processes. This creates a complex global patent expiry calendar that pharmaceutical companies must meticulously track and strategize around.
The rise of biosimilars, the generic equivalents of biologic drugs, adds another layer of complexity to this landscape. Biologics, which are complex protein-based medications, have different manufacturing processes and are generally more expensive than traditional small-molecule drugs. The patent landscape for biologics is also more intricate, with longer development times and complex regulatory pathways for biosimilar approval. However, as biosimilar competition emerges, it is expected to follow a similar trajectory of increasing affordability and market disruption for originator biologics.
The current wave of patent expiries is particularly noteworthy due to the sheer volume and revenue generated by the drugs involved. These are not niche medications but often widely prescribed treatments for chronic conditions, infections, and other significant health concerns. The economic ripple effect extends beyond pharmaceutical companies to include healthcare payers, pharmacy benefit managers, and ultimately, patients who stand to benefit from reduced medication costs.
Looking ahead, the pharmaceutical industry will continue to navigate the cyclical nature of patent expiries. Companies that prioritize innovation, build robust and diversified pipelines, and adapt their business models to embrace both branded and generic strategies are likely to fare best. The ongoing challenge lies in balancing the need for profitability, which funds future innovation, with the societal imperative for accessible and affordable healthcare. The approaching patent cliff is a stark reminder of this delicate equilibrium and the dynamic forces shaping the future of medicine.