The Indian government is poised to implement significant amendments to its regulatory framework governing blood products and plasma-derived medicinal products (PDMPs), a strategic move aimed at streamlining manufacturing processes, reducing costs, and enhancing the availability of life-saving therapies. These proposed changes, detailed in a draft notification by the Ministry of Health and Family Welfare on March 9, target the elimination of redundant viral testing on finished plasma products, bringing India’s protocols into closer alignment with established international pharmacopoeial standards. This shift marks a crucial step in modernizing the nation’s biopharmaceutical sector, promising substantial benefits for both patients and the burgeoning domestic industry.
Plasma-derived medicines represent a vital class of biological therapeutics essential for treating a wide array of severe and often chronic conditions. Products such as albumin are indispensable for managing trauma, burns, and liver diseases; immunoglobulins are critical for patients with primary immune deficiencies, autoimmune disorders, and severe infections; and clotting factors are life-saving for individuals with haemophilia and other bleeding disorders. The global demand for these therapies has been steadily climbing, driven by an aging population, improved diagnostics, and increasing prevalence of chronic illnesses. India, despite its vast population and growing healthcare needs, remains heavily reliant on imports for these critical products, with over 60% of its plasma-derived product requirements currently met by foreign manufacturers. This dependency underscores the urgency of bolstering domestic production capabilities and reducing supply chain vulnerabilities.
The core of the proposed regulatory overhaul lies in discontinuing the "double testing" requirement for viral markers such as HIV (Type I and II antibodies), Hepatitis B surface antigen, and Hepatitis C virus antibodies on finished plasma products. Currently, manufacturers are mandated to conduct these tests at two distinct stages: first, on the raw source plasma, and then again on the final, manufactured medicine. The government’s argument, backed by global scientific consensus, is that once the source plasma has been rigorously verified for safety according to stringent international standards, repeating the identical tests on the finished product adds unnecessary layers of complexity, time, and cost without any demonstrable enhancement in product safety. Modern manufacturing processes for PDMPs incorporate multiple robust viral inactivation and removal steps, which are highly effective in neutralizing potential viral contaminants, rendering subsequent re-testing of the final product largely superfluous from a safety perspective.
Industry experts and healthcare professionals have largely welcomed this forward-thinking approach. Dr. Harpreet Kaur, Medical Superintendent and Head of Lab Services and Blood Centre at Aakash Healthcare in Delhi, emphasized the dual benefits, stating, "The process of production of the product involves multiple viral inactivation processes, and the blood products undergo testing at the donor level and pre-processing. The second testing causes delays and adds to the cost of production." She further highlighted the economic rationale, asserting that "rational and cost-efficient manufacturing will help support domestic manufacturers, which in turn will improve availability of immunoglobulins, clotting factors and other essential therapies." This perspective underscores the anticipated domino effect: reduced production hurdles will translate into more affordable and accessible medicines for patients across the country.

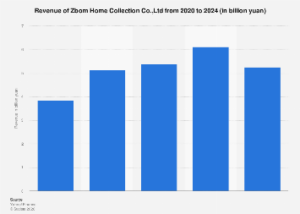
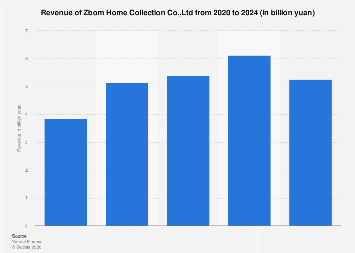
Economically, the impact of this regulatory streamlining is expected to be substantial. The Indian plasma-derived products market, valued at approximately $383.5 million in 2024, is projected for rapid expansion, with analysts forecasting significant growth by 2032. Eliminating redundant testing could translate into direct cost savings for manufacturers, potentially lowering the final price of these critical medicines. Furthermore, shortening regulatory timelines by removing this step will accelerate the release of product batches, mitigate the risk of medicine shortages, and improve inventory management. This efficiency gain is particularly crucial for products with complex manufacturing cycles and high demand, such as albumin and immunoglobulins, where timely availability can be life-saving. The "Make in India" initiative stands to benefit immensely, as domestic manufacturers will find it more competitive to produce these high-value biologics, reducing the nation’s import burden and fostering self-reliance in a critical healthcare segment.
The decision to rationalize testing protocols brings India squarely in line with leading global pharmacopoeias and regulatory bodies. Standards set by the Indian Pharmacopoeia (IP), British Pharmacopoeia (BP), United States Pharmacopeia (USP), and European Pharmacopoeia (EP) all prioritize comprehensive viral screening at the "first homogeneous pool" stage of raw plasma. This internationally accepted methodology focuses on ensuring the safety of the bulk raw material before it undergoes fractionation and purification into various therapeutic proteins. By adopting this globally recognized best practice, India not only enhances its regulatory credibility but also facilitates easier market access for Indian-manufactured PDMPs in international markets, potentially opening avenues for exports and further boosting the domestic biopharmaceutical industry.
However, the success of this regulatory shift hinges critically on the robustness of the initial donor screening and plasma collection infrastructure. As Dr. Kaur rightly pointed out, "what needs to be strengthened is the quality of donor testing across pan-India blood centres, ensuring NAT testing for all, and ensuring full traceability of plasma units." Nucleic Acid Testing (NAT) for HIV, Hepatitis B, and Hepatitis C is considered the gold standard for donor screening, offering a much shorter window period for detection compared to traditional antibody or antigen tests. Mandating universal NAT testing and establishing robust traceability systems for every plasma unit collected are paramount to maintaining the highest safety standards at the source. This comprehensive approach, combined with stringent adherence to Good Manufacturing Practices (GMP) and effective viral inactivation steps during processing, forms a multi-layered safety net that justifies the elimination of redundant end-product testing.
The proposed amendments also underscore the dynamic nature of public health and the need for adaptable regulatory frameworks. While current measures adequately address known viral threats, continuous vigilance and a preparedness to address emerging viral diseases are essential. Future challenges, such as novel pathogens, might necessitate additional testing protocols, but these would be introduced based on evolving scientific evidence and risk assessments, rather than blanket, redundant testing. Maintaining public trust in the safety of plasma-derived medicines is paramount, and transparent communication about the enhanced initial screening processes and the multi-layered safety protocols will be key to ensuring public confidence.
Beyond the immediate benefits for PDMP manufacturers and patients, this regulatory evolution signals India’s broader ambition to solidify its position as a global pharmaceutical and biopharmaceutical hub. By embracing international best practices and streamlining its regulatory environment, India enhances its attractiveness for foreign investment in the biopharma sector, stimulates innovation, and fosters a more competitive domestic landscape. This strategic realignment is not merely about removing a testing requirement; it represents a philosophical shift towards a risk-based, scientifically informed regulatory approach that prioritizes efficiency without compromising safety. As the Ministry of Health invites public comments on the draft notification, the stage is set for a transformative chapter in India’s healthcare and pharmaceutical journey, promising greater accessibility to life-saving medicines and a stronger, more globally integrated biopharmaceutical industry.